Guidelines for Packaging, Shipping and Transport to OSPHL
Newborn Screening Samples
For information about mailing Newborn Screening samples only, see the Newborn Screening Practitioner's Manual (pdf).
Courier Service
The OSPHL Courier Handbook contains packaging instructions for specimens picked up by OSPHL couriers.
Guidelines for Non-Courier Packaging and Shipping
View guidelines.
Emergency Transport
For emergency transport of samples to OSPHL, call 503-693-4100 (Monday-Friday 8am to 5pm); or 971-673-1111 (after hours).
Shipping Methods
Specimens can be shipped to OSPHL via courier, mail, FedEx or UPS following appropriate regulations.
Note: Category A shipments cannot be transported by USPS or UPS.
Ship specimens to:
Oregon State Public Health Laboratory
7202 NE Evergreen Parkway, Suite 100
Hillsboro, OR 97124
Phone: 503-693-4100
Training and Resources
Appropriate regulations for the shipment of infectious materials must be followed when sending specimens to OSPHL. The shipper is responsible for the proper packaging and shipping of all specimens. Shippers must be trained and certified by their employer to be able to prepare and ship packages containing category A and B infectious substances.
- Specimens or cultured organisms transported by ground, air, rail, and water are subject to the regulation of the Department of Transportation (DOT).
- Specimens transported by air are also subject to the regulations of the International Air Transport Association (IATA).
Packaging and Shipping Training
Resources for Shipping Regulations
For questions or assistance with shipping and transport contact OSPHL: 503-693-4100.
Packaging and Shipping Guidelines (Non-Courier)
- Categorize the agent being shipped (Category A, Category B).
Refer to  DOT brochure "Transporting Infectious Substances Safely" if guidance is needed.
DOT brochure "Transporting Infectious Substances Safely" if guidance is needed.
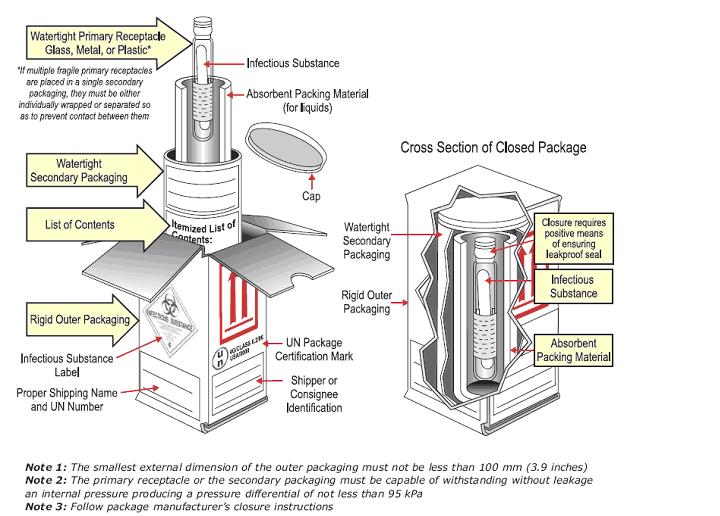
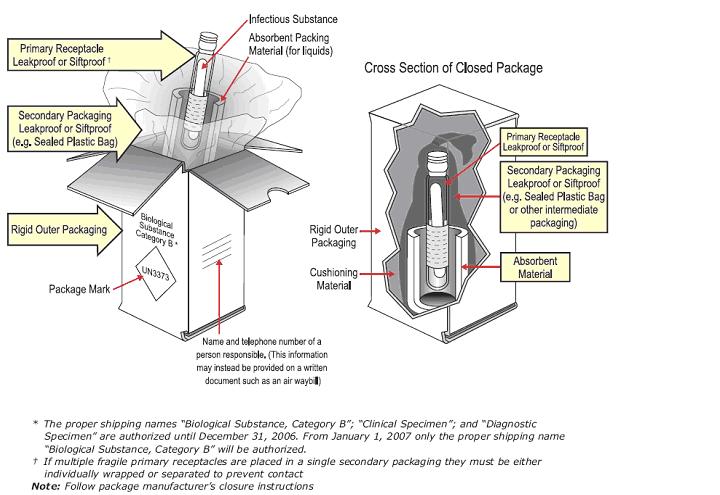
- Package sample (Category A, or B) using "triple packaging"
NOTE: This is a general description for packaging. Refer to the manufacturer’s instructions for use that was received with the packaging materials for specific guidelines. The manufacturer’s instructions must be followed by the shipper, or the package is not acceptable for transport. Packaging materials from different vendors can’t be combined.
- A leak-proof primary receptacle(s) such as a sealed test tube, etc.
- A leak-proof secondary packaging
- Sufficient absorbent placed inside the secondary packaging to fully absorb the specimen volume if the primary receptacle(s) leaks or breaks. Note: if the specimen is solid, no absorbent is necessary.
- Outer packaging of adequate strength for its capacity, weight and intended use.
- Complete a "Shipper’s Declaration" form for Category A substances.
Note: Shipper’s Declarations are not required for UN3373 Biological substance, Category B.
- Forms are available at the shipping site and can be completed by a staff member trained to do packaging and shipping.
- Two copies of the declaration are shipped with the sample.
CATEGORY A EXAMPLE:
CATEGORY B EXAMPLE:
NOTE 1: At least one surface of the outer packaging must have a minimum dimension of 100mm x 100mm (3.9 inches).
NOTE 2: For liquid shipments by aircraft, the primary receptacle or the secondary packaging must be capable of withstanding leakage an internal pressure producing a pressure differential of not less than 95 kPa.