Administrative Rule changes made in January 2026 clarify what products [link the word “products” back to the Veterinary Products page] need to be registered. They also added requirements to what information is recorded at registration in order to assist inspectors in identifying registered vs unregistered products. This information will need to be provided at renewal if not recorded during a prior registration.
Brand and Product Name requirements
Record the complete brand and product names as they appear on the label. Abbreviations used during registration that are not used on the product itself may cause products to be deemed unregistered during inspection.
Formulation and Concentration
Formulation examples include tablet, chew, liquid, gel, pellet, etc. The concentration or strength of the product must be indicated when present. This could be a specific drug concentration or a general statement such as “extra strength” or “large breed” if such wording is used on the label.
UPC lists
As of January 2026, ODA requires UPCs displayed on product labels to be kept on file for all products. Multiple UPCs can be listed under the same registration only when identical products are offered in multiple package sizes or types and must include distinguishing factors. UPCs can be updated online during renewal or at any other time by email. Products sold without UPCs should be noted as such.
What are “distinguishing factors”?
Distinguishing factors are used to indicate how multiple UPCs apply to the same product. Most often, they describe package count or size when an identical formula is offered in multiple package sizes, or when the packaging style is updated and a new UPC is assigned. These can be as simple as “5# bag” and “50# bag”.
When to Separate or Combine Products
What items can be registered together?
Items that are offered in multiple package types or sizes, where the brand and product names, formula, and all other labeling is identical may be registered together.
- Quantity or count differences:
- 12 oz & 128 oz bottles
- 5# bag & 40# bags
- 30-count and 100-count bottles
- single-dose & 6-packs, etc.
- Packaging material changes, such as switching from tubs to bags
- Products sold under multiple SKUs at different distributors, but all product information and the formula are identical.
How do I tell when products require individual registrations?
- Individual registrations are required for any product readily distinguishable from another by content, brand name, trade name, manufacturer, use as specified in labeling, formulation, concentration, dosage form, or other distinction. Examples include:
- Products offered in:
- Different strengths or concentrations, such as 25 mg vs 100 mg, 50 mg/mL vs 100 mg/mL, or regular vs maximum strength
- Multiple formulations or dosage forms, such as tablets, chews, pellets, powders, crumbles, etc.
- Multiple flavors or scents
- An item sold under multiple “white label” brand names. Each brand name requires separate registration.
- A product line offering multiple size options, where the label specifies use by specific sizes, weight ranges, ages, etc (i.e. small, medium, large or listing specific weight ranges on each package).
- Items sold in different presentations and containing different package directions, such as a dewormer sold in a single dose with one set of instructions (“use once” or “use once every 3 months”) and also packaged as a 7-day course consisting of 7 doses and different instructions for use (“use daily for 7 days in a row”).
- Products whose brand or product names have changed. The new product must be registered prior to release, and the old product must be registered as long as it is still in distribution/offered for sale.
Other Common Questions
Name changes and discontinued items
All products offered or exposed for sale in Oregon must be registered, even if they have been discontinued or the brand or product name is changing. Older products will require separate registrations until the old stock has been sold or removed from distribution entirely. If a discontinued product or former product name can be found in distribution in Oregon, it must be registered.
Are “ 25b” pesticides exempt from Animal Remedy registration because they’re registered with the ODA Pesticides Program?
No. Only products that are regulated (registered) under the Federal Insecticide, Fungicide, and Rodenticide Act are exempt. If the 25b product makes health claims, it must be registered.
Sample Registration
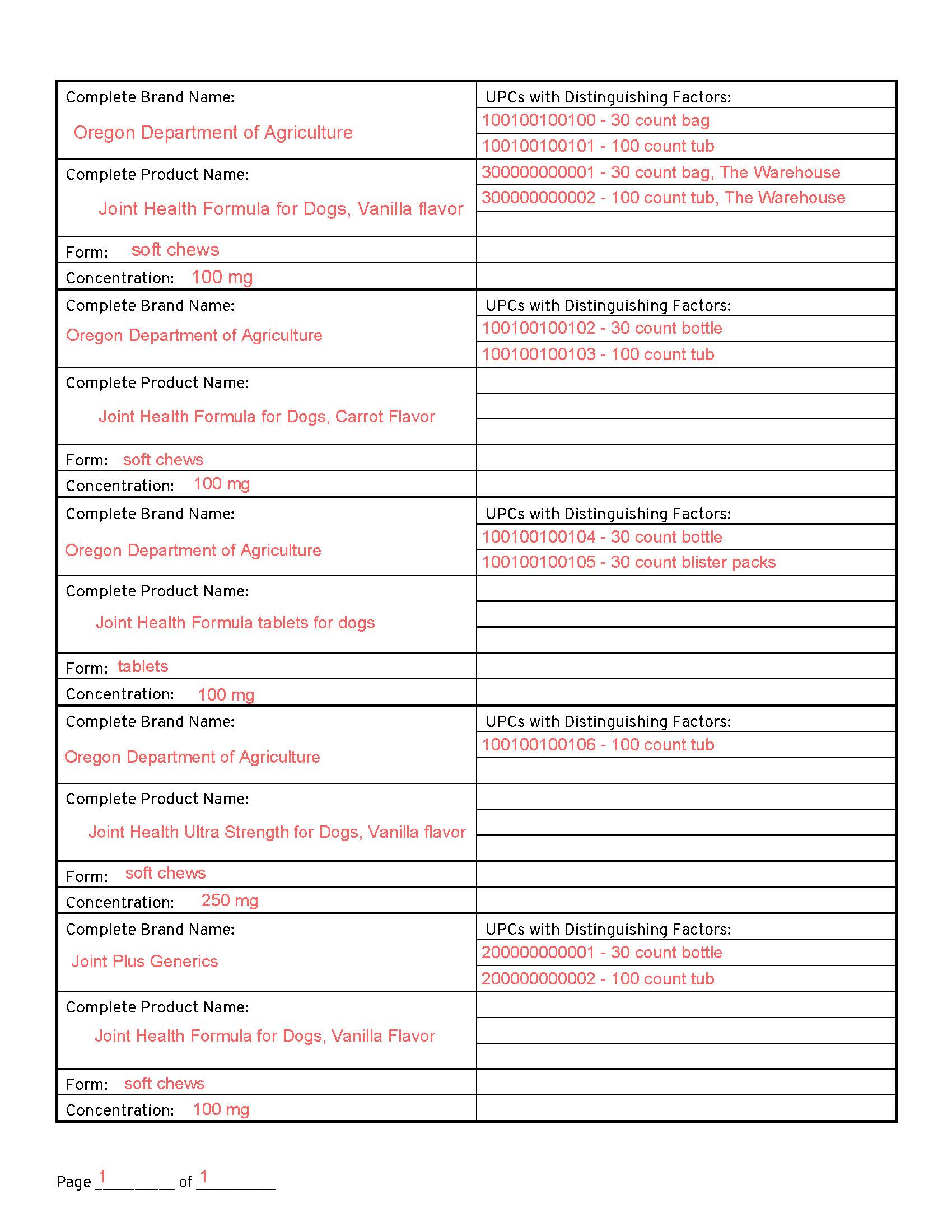
- All package sizes of Vanilla Flavor are registered under 1 registration, listing UPCs for package variations. “The Warehouse” store uses different UPCs but all other product details are the same, so those UPCs are registered under item 1 as well.
- All package sizes of Carrot flavor are registered separately, listing UPCs for package variations.
- Tablets are registered separately, as a distinctly different form than the chews. The company switched from blister packs to bottles, so they list both UPCs available in the marketplace.
- Ultra Strength, though also vanilla flavor, is a different concentration than the 100 mg product and requires separate registration.
- Joint+ Generics is a distinctly different brand name or “white label” and must be registered separately, even though it otherwise shares the same description as #1.