Background & Introduction
The information on this page is intended for operators and owners of water systems that treat a surface water source. Water systems that treat surface water sources have to deal with complicated
regulations for Oregon Drinking Water Services (DWS),
Department of Environmental Quality,
Water Resources Department, and various other state and local entities.
In addition to these regulatory requirements, systems have to adhere to
National Sanitation Foundation treatment and
 Oregon's construction standards,
water rights requirements, constantly changing raw water quality including periodic
algae blooms and high
Oregon's construction standards,
water rights requirements, constantly changing raw water quality including periodic
algae blooms and high
 turbidity events, and costly management of various assets. Information below is being presented to provide operators the information and tools needed to optimize water treatment processes to maximize public health protection without costly capital improvements.
turbidity events, and costly management of various assets. Information below is being presented to provide operators the information and tools needed to optimize water treatment processes to maximize public health protection without costly capital improvements.
Although primarily focused on treatment optimization, this page also provides
technical,
managerial, and
financial resources (e.g., Oregon's
Drinking Water State Revolving Fund) needed to build water system capacity. Particularly useful is the U.S. Environmental Protection Agency's
Small Public Water Systems and Capacity Development Web site and the free technical assistance offered through the DWS
Circuit Rider Program. If you have specific topics that you believe should be included on this page, comments about the content of this material, or discover links that do not work, please
contact us.
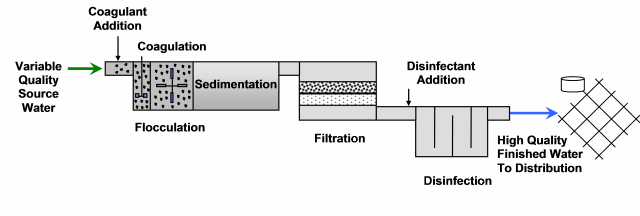
Figure 1. Multiple Barriers
Water Treatment Optimization
Water treatment plant optimization is the process of improving the performance of particle removal beyond regulatory requirements without making major capital expenditures. As outbreaks (e.g. in 1993 in Milwaukie, WI) have made clear it clear that just meeting current regulatory water quality standards might not be enough to protect against contaminants. For these reasons, surface water treatment plants are encouraged to optimize their treatment processes and adopt more stringent
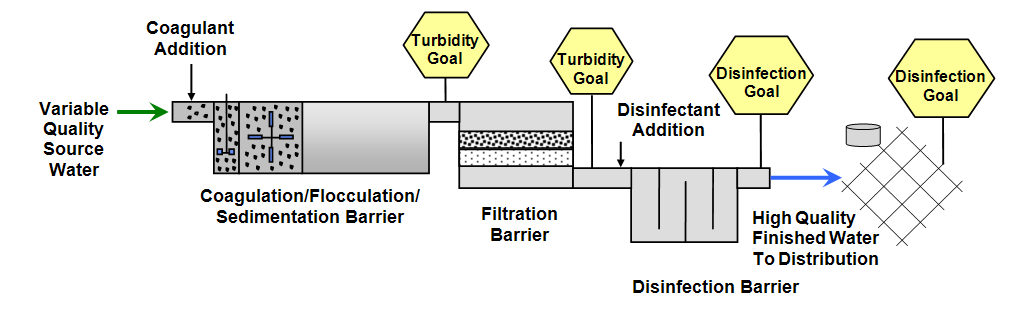
 water quality goals
water quality goals as a component of the
 multiple barrier approach
multiple barrier approach to protect against these contaminants and prevent waterborne disease outbreaks. For more information and a table of recommended optimization goals, read about Area Wide Optimization below.
Figure 2. Multiple Barrier Goals
Area Wide Optimization (AWOP)
In order to assist treatment plant operators and promote optimization concepts, a network of partners including the
U.S. Environmental Protection Agency (EPA), state drinking water programs, Process Applications, Inc., and the
Association of State Drinking Water Program Administrators (ASDWA) have teamed up to implement a national
Area Wide Optimization Program (AWOP). AWOP is a non-regulatory approach to identify and address performance limiting factors at surface water treatment plants.
DWS has joined with neighboring states and
EPA Region 10 to participate in AWOP. In Oregon, AWOP is focused on optimizing particle removal at existing surface water treatment plants in order to maximize public health protection by minimizing exposure to pathogens such as
 Giardia
Giardia and
Cryptosporidium.
Oregon's AWOP is currently focusing on implementing optimization activities for
conventional and direct filtration treatment plants and establishing goals for
 turbidity
turbidity removal. Conventional treatment includes a clarification process prior to filtration. In direct filtration, coagulated water is directed straight to the filters without the intermediate clarification process. The diagrams below illustrate the differences between conventional and direct filtration:
Figure 3. Conventional Filtration Schematic
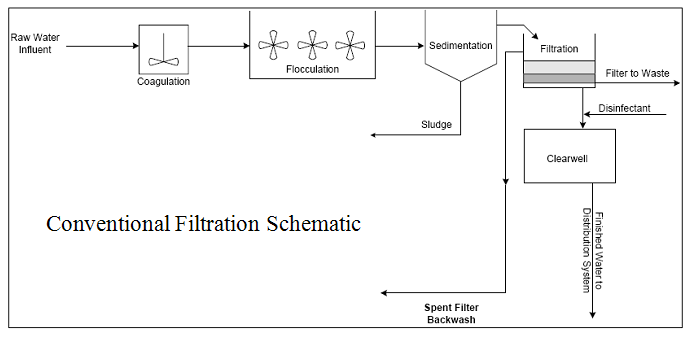 Figure 4. Direct Filtration Schematic
Figure 4. Direct Filtration Schematic
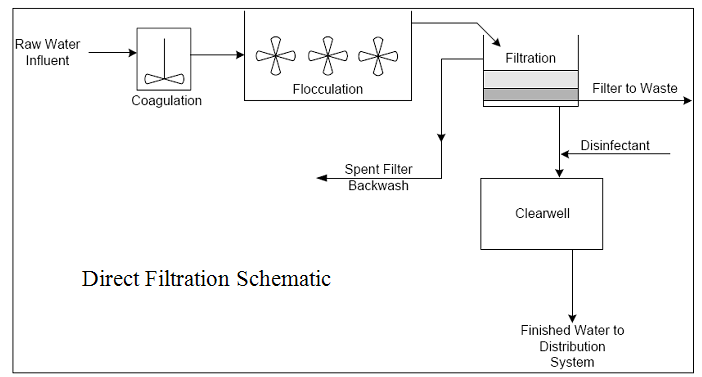 Table 1. Oregon Area Wide Optimization (AWOP) and Water Treatment
Table 1. Oregon Area Wide Optimization (AWOP) and Water Treatment
Plant Goals are listed below (refer to water treatment and system type)
SEDIMENTATION
(for Conventional Filtration) |
Turbidity |
Criteria |
| Settled Water | = 2.0 NTU, 95% of the time. | If average annual raw water turbidity is > 10 NTU. |
| = 1.0 NTU, 95% of the time. | If average annual raw water turbidity is = 10 NTU. |
FILTRATON
(for Conventional and Direct Systems) |
Turbidity |
Criteria |
| IFE and CFE Filtered Water | - Turbidity = 0.10 NTU, 95% of the time.
- Max. turbidity = 0.30 NTU.
| Based on maximum values recorded during 4-hour increments (excluding the 15-minute period following backwash). |
| IFE filtered water after backwash | - Turbidity returns to = 0.10 NTU within 15 minutes after backwash.
- Max. spike = 0.30 NTU.
- Turbidity at return to service = 0.10 NTU.
| Goals apply to both systems with and without filter-to-waste capability. Goals also apply to the backwash recovery period starting immediately after backwash. |
| IFE = Individual Filter Effluent; CFE = Combined Filter Effluent |
For more information, click links below:
Coagulation
 Coagulation
Coagulation is a process where chemicals are added to water in order to improve subsequent treatment processes. Chemicals may include pH adjusters and various polymers. Coagulants (such as alum) neutralize positive or negative charges on small particles allowing them to stick together and form larger particles that are more easily removed by sedimentation (i.e., settling) or filtration. Chemicals and components used to treat water must be certified for potable use under
NSF/ANSI Standard 60 (chemicals) or 61 (components).

A variety of devices, such as baffles, static mixers (shown right), turbines (shown extreme right), impellers, and in-line sprays can be used to mix the water and distribute the chemicals evenly. This process is usually called rapid mix.
Click the links below to learn how to:
Flocculation
 Flocculation
Flocculation typically follows coagulation and rapid mixing, and is the process where chemically treated water is sent into one or more basins (or stages) where suspended particles can collide and agglomerate (i.e., stick together) and form larger particles called "floc." Gentle agitation of the water by paddle mixers (shown right) or impeller type blades and appropriate detention times (i.e., the length of time water remains in the basin) help facilitate this process.
It is important that conventional filtration systems with a treatment plant sedimentation process strive for a larger, heavier floc that is easier to settle out. In some types of treatment systems, such as direct filtration without a sedimentation/clarification process, a smaller "pin" floc is more desirable.
Sedimentation/Clarification

Following
 flocculation
flocculation, a
 sedimentation or clarification
sedimentation or clarification step can be used to remove larger particles. During sedimentation, the velocity of the water is decreased so that the suspended material (including flocculated particles) can settle out by gravity. Once settled, the particles combine to form a sludge that is later removed from the bottom of the basin. Settling plates (shown right) or incline square or rectangular tubes (shown below right) are sometimes used to accelerate this process. A wide variety of other clarification processes can then be used to remove floc as well. Sedimentation used to remove solids prior to chemical addition (called pre-sedimentation) may also be employed to aid treatment.

Learn more about the following
 topics
topics:
- Dissolved Air Flotation (DAF),
- Contact adsorption clarifiers,
- Solids contact clarifiers, and
- Optimize sedimentation.
Filtration

With most of the larger particles typically removed after
 sedimentation/clarification
sedimentation/clarification, clarified water passes through a filtration process. In rapid sand filtration, at a rate of between 2-10 gpm per square foot, the water is filtered through an approximate 36-inch depth of graded sand. Filters are periodically cleaned by backwashing (reversing the flow of water through the filter). Anthracite coal or activated carbon may also be included in addition to sand to improve the filtration process, especially for the removal of organic contaminants and taste and odor problems. Pressure filters are similar to rapid sand filters, except that the water enters the filter under pressure. Slow sand filtration occurs at a slower rate of 0.5-1.0 gpm per square foot and is aided by a biological layer called schmutzdecke.
Other types of filtration processes can be used without coagulation, and include membrane and cartridge filtration, as well as diatomaceous earth.
Click the links below to learn how to:

Click the links below to learn more about:
Disinfection

 Disinfection
Disinfection involves inactivating pathogens from filtered water with the use of chlorine, chloramines, or other oxidizing agent, and can include ultraviolet light.
 Chlorine disinfection
Chlorine disinfection is achieved through delivering a chlorine dose with adequate contact time (CT) (shown right). The parameter CT is defined as the free Chlorine residual as measured at or before the first user (in mg/l) multiplied by the contact Time (in minutes) between the point of chlorine addition and the point at which chlorine is measured. [CT = Chlorine concentration X contact Time]
 Chloramination
Chloramination involves adding ammonia to chlorinated water. Although not as effective a disinfectant, chloramination allows the chlorine residual to be carried further in large distribution systems, and has the added benefit of reducing the formation of disinfection byproducts (a product of the reaction of chlorine with organic matter).
 Ozone
Ozone is a powerful oxidizing and disinfection agent, and is formed by passing dry air through a system of high voltage.
With
 ultraviolet light
ultraviolet light (shown below right), there is no residual concentration to measure in the water. Other parameters such as UVT, intensity, dose, and flow are used to verify adequate disinfection for systems with ultraviolet light.

Click the links below to learn more about:
Forms & Tools
The following links will direct you to forms and tools for use by surface water treatment operators. These resources include public notice templates, USEPA quick reference guides for surface water treatment rules, turbidity and coliform sampling requirements, and the monthly operating report (MOR) forms used to report turbidity and disinfection data.